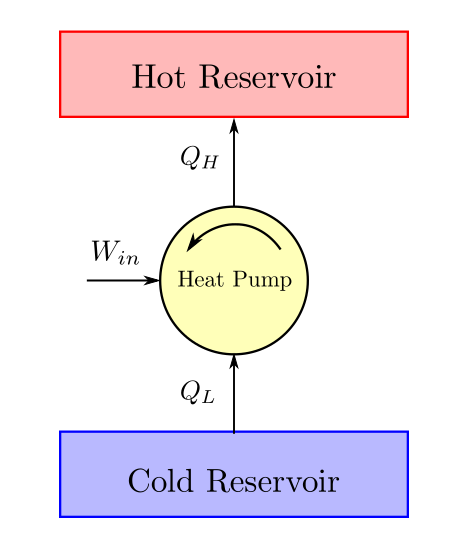

For example, the electrical energy going into a refrigerator is converted to heat and goes out the back, representing a net increase in entropy. This can happen in a non-isolated system if entropy is created elsewhere, such that the "total" entropy is constant or increasing, as required by the second law. Note that from the mathematical definition of entropy, a process in which heat flows from cold to hot has decreasing entropy. For example in a refrigerator, heat flows from cold to hot, but only when aided by an external agent (i.e. Informally, "Heat doesn't flow from cold to hot (without work input)", which is obviously true from everyday experience. If a system is at equilibrium, by definition no spontaneous processes occur, and therefore the system is at maximum entropy.Īlso, due to Rudolf Clausius, is the simplest formulation of the second law, the heat formulation or Clausius statement: Heat generally cannot spontaneously flow from a material at lower temperature to a material at higher temperature. (An exception to this rule is a reversible or "isentropic" process, such as frictionless adiabatic compression.) Processes that decrease total entropy of the universe are impossible. Thus, while a system can undergo some physical process that decreases its own entropy, the entropy of the universe (which includes the system and its surroundings) must increase overall. The formulation of the second law that refers to entropy directly is as follows: In a system, a process that occurs will tend to increase the total entropy of the universe. Thus, the theorems of thermodynamics can be proved using any form of the second law and third law There are many ways of stating the second law of thermodynamics, but all are equivalent in the sense that each form of the second law logically implies every other form. There are many versions of the second law, but they all have the same effect, which is to explain the phenomenon of irreversibility in nature.

Entropy is a measure of how far along this evening-out process has progressed. In simple terms, the second law is an expression of the fact that over time, ignoring the effects of self-gravity, differences in temperature, pressure, and density tend to even out in a physical system that is isolated from the outside world. The second law traces its origin to French physicist Sadi Carnot's 1824 paper " Reflections on the Motive Power of Fire", which presented the view that motive power ( work) is due to the fall of caloric ( heat) from a hot to cold body ( working substance). But the entropy of a pure substance is usually given as an intensive property - either entropy per unit mass (SI unit: J K-1 kg-1) or entropy per unit amount of substance (SI unit: J K-1 mol-1).The second law of thermodynamics is an expression of the universal law of increasing entropy, stating that the entropy of an isolated system which is not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium. It has the dimension of energy divided by temperature, which has a unit of joules per kelvin (J K-1) in the International System of Units (or kg m2 s-2 K-1 in basic units). Entropy was discovered when it was noticed to be a quantity that behaves as a function of state, as a consequence of the second law of thermodynamics.Įntropy is an extensive property. The concept of entropy has been found to be generally useful and has several other formulations. (If heat is transferred out the sign would be reversed giving a decrease in entropy of the system.) The above definition is sometimes called the macroscopic definition of entropy because it can be used without regard to any microscopic description of the contents of a system. Where T is the absolute temperature of the system, dividing an incremental reversible transfer of heat into that system (dQ).
